A Buyer’s Playbook for Resistant Dextrin + MCC in the GLP‑1 Era
Fiber has moved from "nice to have" to a frontline lever in product renovation—especially as the GLP‑1 conversation pushes brands to rethink satiety, metabolic positioning, and what consumers will tolerate day after day. Procurement teams are feeling this shift in a very specific way: demand is rising for resistant dextrin (and related soluble fibers) in foods and supplements, while microcrystalline cellulose (MCC) remains a quiet workhorse for solid dosage formats that often sit next to weight-management or metabolic-health portfolios.
This article is written for buyers who need a repeatable sourcing method—not a one-off supplier story. It focuses on how to define specs, avoid labeling pitfalls in the U.S. and EU, and evaluate a Recommended Chinese Resistant Dextrin Manufacturer alongside a Recommended Chinese Microcrystalline Cellulose Manufacturer—without letting "good price" turn into downstream compliance and quality cost.

1. Why the GLP‑1 Era is Changing Fiber Procurement
Two realities are playing out at the same time in the functional ingredient market:
- Fiber is becoming a primary innovation theme, not just a nutrition panel upgrade.
- Product teams want ingredients that support low net carbs, low calorie density, and consumer-friendly tolerance.
A recent market overview highlighted how fiber is positioned to dominate food-and-beverage health trends heading into 2026, with GLP‑1 adoption amplifying interest in satiety and blood sugar management. In practical procurement terms, that "trend" translates into longer RFQ lists for resistant dextrin, resistant maltodextrin, and other low-calorie soluble fibers.
At the same time, MCC sourcing often runs in parallel—because many brands launch fiber-forward concepts across both:
- Foods & beverages (where resistant dextrin is the star)
- Tablets/capsules (where MCC helps the format succeed at scale)
If resistant dextrin supports the serving-level fiber target and sensory goals, MCC frequently supports manufacturing stability—flow, compressibility, and consistent disintegration—especially in high-output tablet lines.
2. Ingredient Fundamentals That Matter to Buyers
This section stays buyer-oriented: what the ingredient is, how it behaves, and what must be verified on paper and in incoming QC.
Resistant Dextrin: What to Know Before You Approve a Spec Sheet
Resistant dextrin is typically positioned as a soluble dietary fiber that resists digestion in the small intestine and is fermented in the large intestine. For low-carb and keto-positioned products, resistant dextrin is commonly treated as non-net-carb contribution in formulation logic because it is indigestible.
From a procurement perspective, resistant dextrin is attractive because it can be specified around:
- Neutral taste and low interference with existing flavor systems.
- Low viscosity and easy processing in many beverage and powder systems.
- Fiber-forward positioning relevant to resistant dextrin GLP-1 weight management concepts (satiety/glycemic management narratives often sit adjacent to GLP‑1 conversations).
A commonly used buyer-facing baseline spec set includes:
| Buyer-relevant parameter | Why it matters in procurement | Typical declaration seen in market materials* |
|---|---|---|
| Fiber content | Determines cost per gram of deliverable fiber | ≥82% fiber |
| Appearance | Affects finished product color and consumer perception | White to light yellow |
| Protein | Quality/consistency signal for starch-derived fibers | ≤6.0% |
| Storage | Impacts shelf-life planning and warehouse handling | Store in a cool place |
Buyer note: resistant dextrin is sometimes used interchangeably in commercial discussions with resistant maltodextrin or digestion-resistant maltodextrin. Don't accept interchangeability casually—lock the exact naming, test methods, and labeling basis into your specification.
MCC: Why “Pharma Excipient” Sourcing is Not the Same as Food Ingredient Sourcing
Microcrystalline cellulose (MCC) is a widely used excipient in tablets and capsules, often serving as filler, binder, disintegrant support, or flow aid.
For procurement teams working with a Recommended Chinese Microcrystalline Cellulose Supplier, the key is not just whether MCC is available—but whether documentation matches the intended pharmacopeial and customer requirements.
A common buyer anchor is the USP MCC monograph, which outlines official quality expectations and harmonization work. In practice, MCC purchasing should be treated as a compliance-managed excipient buy, with COA alignment, change control discipline, and lot-to-lot consistency checks.
3. Regulatory Fit: Labeling Discipline Affects What You Buy
Regulatory alignment is not "legal's problem later." If the spec and documentation are wrong, you can end up with inventory you can't label—or can't ship.
U.S. Labeling: Dietary Fiber Declaration Isn't Optional
In the U.S., the FDA has issued guidance on declaring certain isolated or synthetic non-digestible carbohydrates as dietary fiber, including resistant maltodextrin. Procurement teams must:
- Confirm the ingredient identity and labeling basis your regulatory team expects.
- Require a COA and specification package that clearly supports the ingredient's dietary fiber declaration for U.S. Nutrition/Supplement Facts use.
- If your product concept is built around resistant dextrin prebiotic benefits, make sure the "fiber" basis is documented correctly for your market.
EU Discipline: Claims Must Stay Narrow
In the EU context, health claims can't drift into broad, consumer-friendly language unless the wording and conditions are appropriate. If your commercial team is planning an EU rollout, align with regulatory early so that your resistant dextrin spec, identity, and substantiation package support what marketing wants to say.
4. Formulation Solutions Buyers Should Expect to See
Resistant dextrin and MCC are often evaluated separately. In real product pipelines, they show up together in portfolio planning: powders and beverages drive resistant dextrin volumes; tablets/capsules keep MCC on the approved-vendor list.

Low-Carb Confectionery: Bulk + Fiber Without Texture Penalties
In confectionery and "better-for-you" sweets, resistant dextrin is commonly chosen to help increase fiber without dramatically changing flavor and support sugar-reduction strategies. Where buyers get surprised is not functionality—it's tolerance and serving-size realism. If you're purchasing resistant dextrin for confectionery, make sure the supplier can support consistent fiber content and clear microbiological controls.
Diet Powders: The “Quiet” Fiber
Powdered nutrition formats are one of the cleanest volume channels for resistant dextrin because they can absorb meaningful grams of fiber per serving. Buyer-facing expectations for resistant dextrin in powders include easy dissolution in water, a neutral taste profile, and low viscosity so the drink doesn't feel "thick" at higher fiber loads.
MCC in Tablet Formulation: Preventing “Compression Surprises”
When MCC is used in tablets, procurement often inherits the consequences of small changes in particle size distribution, moisture content, or bulk density. Those changes can affect blending, weight variation, and tablet hardness. That's why MCC sourcing should be managed like a controlled excipient program—requiring current monograph-aligned COAs and strict change notification expectations.
5. What to Verify When Sourcing Resistant Dextrin from China
The fastest way to reduce sourcing risk is to run a consistent checklist across every Resistant Dextrin Supplier China candidate. A BRC-style audit checklist can be a helpful template for food ingredient supplier qualification.
Documentation You Should Request
For resistant dextrin, request a document pack that includes:
- Specification sheet (with fiber content target, appearance, protein, moisture, and key quality limits).
- COA template and at least one recent lot COA.
- GMO statement (many buyers specifically require non-GMO positioning for resistant dextrin).
- Allergen statement and contaminant controls as applicable to your market.
Process Capability: Defining “Automation”
Many suppliers describe "fully automated" production. Procurement should translate that into verifiable questions. Is there central control from raw material feeding through filling/packaging? How is lot traceability maintained from starch input to finished resistant dextrin output?
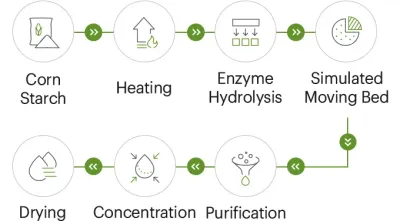
A useful procurement discipline is to require suppliers to map their process controls to your critical-to-quality attributes. If your finished product depends on low viscosity and neutral taste, ask which control points protect those outcomes.
Quality Control and Packaging
A "QC laboratory" line on a brochure is not enough. For resistant dextrin, ask how the supplier verifies fiber content (method and frequency) and microbiological limits. For MCC, the QC expectation is even stricter because it is tied to pharmacopeial compliance.
Furthermore, even high-quality resistant dextrin can be damaged by poor packaging or humidity exposure. Ensure packaging is moisture-resistant and fit for long transit, and that lot codes are readable and traceable at the warehouse level.

6. Sourcing Strategy: Shortlisting and Cost
How to Shortlist Effectively
Search phrases like Recommended Chinese Resistant Dextrin Manufacturer are increasingly used by procurement teams to build an initial longlist. The risk is that "recommended" can mean "well-marketed," not "well-controlled."
A stronger shortlisting approach starts with product-market fit (does the supplier show credible experience in your target application?), moves to compliance fit (can they support dietary fiber declaration?), and ends with execution fit (can they consistent ship and document?).
Cost Strategy
In fiber procurement, the visible price is rarely the true cost. A more decision-useful metric is Cost per gram of delivered fiber (not cost per kilogram of powder). For resistant dextrin, fiber percentage directly affects cost-per-serving economics. A resistant dextrin that meets your spec at ≥82% fiber can reduce the amount required per serving to hit label targets.
A Simple Two-Tier Approval Model
To manage risk while keeping speed, implement a two-tier model:
- Tier 1: Qualification approval (Documentation pass, sample evaluation, COA alignment, and initial audit readiness).
- Tier 2: Commercial approval (Pilot order, destination-lab retest plan, and defined change control expectations).
This approach works especially well when resistant dextrin is entering a high-visibility product line influenced by GLP‑1 adjacent positioning, where launch timelines are tight but compliance tolerance is low.
7. A Buyer Playbook for Repeatable Success
Procurement teams that consistently source high-performing resistant dextrin and MCC typically follow a repeatable sequence:
- Trend fit: Confirm resistant dextrin is the right fiber for your format and consumer tolerance strategy.
- Regulatory fit: Lock your labeling and claims discipline early (FDA dietary fiber declaration logic for the U.S.; careful claim wording discipline in the EU).
- Supplier fit: Evaluate each candidate using the same audit-style checklist—documentation, process control, QC capability, packaging, and change management.
When a supplier passes these gates, resistant dextrin becomes a reliable tool for low-calorie fiber fortification rather than a variable commodity. For buyers needing to evaluate portfolios that span both functional fibers and excipients, reviewing established catalogs can be a practical starting point. To explore specific product specifications and supplier capabilities in this sector, you can visit www.sdshinehealth.com.


