How to Vet a Chinese MCC & Resistant Dextrin Supplier (Without Getting Burned)
For international procurement teams, sourcing Microcrystalline Cellulose (MCC) and resistant dextrin from China has become a strategic necessity. The region offers undeniable scale, competitive cost structures, and a mature export mindset. However, the risks remain distinct: uneven specification control, inconsistent documentation, and the dreaded “good sample / bad bulk” scenarios that can quietly erode product performance.
This guide is written strictly from a buyer’s perspective. It cuts through the marketing noise to focus on what actually matters when qualifying a Microcrystalline Cellulose Supplier or a Resistant Dextrin Manufacturer China-side. We look at how to read specifications, how to audit the production process, and how to build a shortlist that justifies the label of a recommended partner.

The First Decision: MCC and Resistant Dextrin Are Not “Similar Powders”
Buyers often group MCC and resistant dextrin together because they both ship as white-to-off-white powders and frequently appear in the same nutraceutical and pharma-adjacent supply chains. However, from a qualification standpoint, they behave very differently:
- MCC is commonly purchased as a pharmaceutical excipient (and used as a food additive). The primary risks involve pharmacopoeial alignment, consistent grade performance (flow, compressibility), and excipient-style documentation.
- Resistant dextrin is typically purchased as a soluble dietary fiber (also known as indigestible or resistant maltodextrin). The key risks here are fiber assay credibility, microbiological control, and reliable Non-GMO/traceability support.
Treating them with the same audit logic is a common mistake. While a buyer should apply a unified supplier qualification framework, the control points must differ.

Key Quality Benchmarks Before Engaging Suppliers
A sourcing team typically loses the most time not during factory visits, but during email rounds caused by unclear specifications. Before requesting quotations, define one internal spec sheet per ingredient and make it the baseline for every candidate.
MCC: Define the Grade, Then Define “Equivalent”
For MCC, the market often references grade families such as PH-101 vs PH-102. Even when suppliers claim equivalency, buyers should require:
- Declared grade (what they officially call it).
- Target particle size distribution and the specific test method.
- Bulk density / tapped density and acceptance range.
- Loss on drying / moisture limits.
- Microbial limits suitable for the intended use.
- A documentation package that supports pharmacopoeial expectations (USP/Ph. Eur. context).
Expert Sourcing Tip: If an MCC supplier says “we can match any grade,” ask for three recent COAs from three different lots for the same grade. Consistency across lots tells you more than a single “perfect” COA.

Resistant Dextrin: Assay Credibility Over Marketing
For resistant dextrin, there is no single global pharmacopoeial monograph that buyers universally rely on, so practical control points usually include:
- Fiber content (commonly ≥82%, with some commercial grades promoted as higher on a dry basis).
- Protein content (commonly controlled at ≤6.0% in typical export-oriented specs).
- Appearance (white to light yellow) and sensory neutrality.
- Moisture / water activity targets aligned with shelf stability.
- Microbiology panel (APC, coliforms, yeast, mold).
- Non-GMO documentation where required by the destination market.
Why China Dominates—and the Shandong Factor
China’s advantage in both categories is structural, built on a deep base of starch processing and carbohydrate ingredient manufacturing. The country has a mature export ecosystem for food ingredients and excipient-adjacent materials, with established industrial clusters that concentrate talent, utilities, and specialized equipment.
For resistant dextrin in particular, buyers frequently evaluate suppliers in the Shandong food additive cluster. This region is known for functional fibers and related low-calorie carbohydrates. This matters because a cluster tends to improve supply resilience: raw material availability, supporting services, packaging vendors, and experienced logistics partners are easier to secure.
However, clusters also increase the need for due diligence. Two factories located 30 minutes apart can produce powders that look identical but behave differently in applications—especially when process control and QC maturity diverge.

Manufacturing Reality Check: Verifying Process Control
Resistant Dextrin: Process Control is the Differentiator
Most resistant dextrin production narratives sound similar—controlled heating, enzymatic treatment, filtration, concentration, drying, packaging. The real differentiator is how much of that narrative is measured, recorded, and repeatable.
When screening a Resistant Dextrin Manufacturer China-side, verify whether the factory can credibly demonstrate:
- Enzyme management: source control, storage conditions, and lot traceability.
- Automation and central control: reduced manual interventions from feeding to filling.
- In-process controls: checks that predict fiber assay stability and solubility performance.
- Batch records: complete, reviewable, and consistent across lots.
A common export-oriented positioning involves the use of imported biological enzymes and a precision production line of German origin, often paired with a workshop described as GMP standard. These claims are valuable only when they translate into documented controls.
MCC: Grade Consistency via Milling and Classification
For MCC, buyers should focus less on marketing buzzwords and more on what drives performance in tablets and capsules:
- Particle size management (milling, classification, and sampling discipline).
- Moisture control (not just the final test, but how drying and ambient control are managed).
- Contamination prevention (material flow, dust control, and cleaning validation concepts).
Because MCC is frequently used as a pharmaceutical excipient, a Microcrystalline Cellulose Supplier must show maturity in documentation, change control, and deviation handling—not merely a clean workshop.
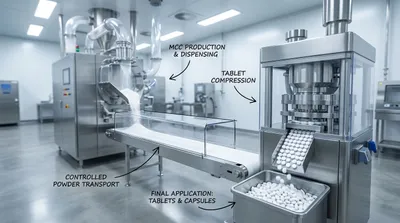
Buyer-Ready Comparison: Standardizing Responses
| Topic | What to confirm with a Microcrystalline Cellulose Supplier | What to confirm with a Resistant Dextrin Manufacturer China-side |
|---|---|---|
| Regulatory Role | Excipient alignment (USP/Ph. Eur. context), grade identity | Food ingredient / soluble fiber positioning, export-ready compliance |
| Core Performance | Flow, compressibility, grade consistency (e.g., PH-family grades) | Fiber assay credibility (often ≥82%), solubility, viscosity behavior |
| Raw Material Control | Pulp/cellulose traceability and qualification | Non-GMO corn/tapioca starch traceability and IP handling |
| Safety Controls | Microbial limits, impurity controls, contamination prevention | Microbiology panel + moisture/water activity for shelf stability |
| Documentation | COA consistency, change control, deviations/CAPA | COA consistency, fiber test method clarity, Non-GMO documentation |
| Site Audit Signals | Data integrity mindset, controlled production environment | Central control/automation, enzyme management, hygienic filling |
The Audit Checklist: Qualifying a “Recommended” Supplier
The term Recommended Chinese Microcrystalline Cellulose Manufacturer (or Recommended Chinese Resistant Dextrin Manufacturer) should mean one thing internally: the supplier can pass a structured audit, deliver stable lots, and support deviations professionally.
1. Company Profile & Compliance Posture
- Legal entity clarity and export history.
- Quality certifications relevant to product type (food safety systems for fibers; excipient-style controls for MCC).
- How the supplier handles customer audits (preparedness often reflects real system maturity).
2. Raw Material Qualification
For resistant dextrin, Non-GMO is not a slogan; it’s a supply-chain discipline. Confirm:
- Approved supplier list and incoming inspection for starch.
- Non-GMO declarations and internal identity preservation (IP) practices.
- Lot mapping: finished product lot → starch lot → supplier batch.
3. Process Control and Automation
For functional fibers, many export-focused factories emphasize a fully automated, centrally controlled workflow. Verify:
- Whether “automation” includes feeding → reaction → drying → filling.
- How manual interventions are recorded.
- Whether critical parameters (temperature, pH, time) are recorded in batch records.
4. QC Laboratory Capability
A lab that can only “confirm pass” is a liability. Buyers should confirm:
- Equipment list relevant to the product (moisture, ash/protein methods, microbiology capability).
- Method discipline: reference methods used for fiber assay, microbial enumeration, and MCC identity tests.
- Retention sample policy and COA review process.
5. Packaging and Logistics
Even good powder can fail if packaging or storage is weak. Confirm:
- Moisture-barrier packaging appropriate for fiber powders.
- Labeling consistency (lot number, manufacturing date, net weight).
- Storage conditions and shelf-life basis.
Application Reality: Proving Performance
A supplier can “meet spec” and still fail your formulation. The fastest way to reduce that risk is to request application-relevant evidence.
- For MCC buyers: Ask the supplier to explain which grades they recommend for direct compression vs. blends, and how they control flow variability across seasons.
- For resistant dextrin buyers: Ask for solubility expectations, handling notes (mixing order, temperature sensitivity), and a realistic view of fiber assay variability across lots. One recurring reason buyers seek a recommended manufacturer is the ability to support stable performance in low-calorie beverages and sugar-reduction formulations.
Cost and Procurement Strategy
For both MCC and resistant dextrin sourcing, experienced buyers evaluate total landed risk, not just the CIF/FOB price. Consider building a simple internal scorecard that weights:
- Quality consistency (COA lot-to-lot stability).
- Deviation responsiveness (speed and quality of investigations).
- Documentation maturity (traceability, change control discipline).
- Logistics stability (packaging, shipment lead times).
The Shandong Supplier Landscape
In the Shandong ecosystem, buyers will find suppliers positioning themselves around Non-GMO corn starch sourcing, imported enzymes, and centralized control. When a supplier can demonstrate these capabilities with evidence—process flow clarity, COA consistency, traceability discipline, and professional audits—it becomes a credible candidate.
As a practical benchmark example for buyers building shortlists, some Shandong-based manufacturers publicly share technical descriptions and product positioning for resistant dextrin and related fibers. One reference point often used during initial market scanning is Shine Health.
Closing Checklist: Shortlist Fast, Validate Deeply
To qualify suppliers efficiently while reducing downstream surprises, procurement teams can follow a two-step approach:
- Shortlist fast (desktop qualification): Require a documentation pack and three recent COAs. Confirm core spec fit and validate Non-GMO claims.
- Validate deeply (audit + trial order): Run a structured site audit using GMP-style thinking. Perform incoming QC on the first three commercial lots and lock in change-control expectations.
Done well, this process lets buyers source confidently from China—ensuring the chosen supplier can sustain quality beyond the first container.


