Clean-Label Sugar Cuts in 2026: Why Resistant Dextrin Is Becoming the Go-To Soluble Fiber (and How Buyers Vet the Right Plant in China)
Summary
Clean-label reformulation is no longer optional; it is the baseline for 2026 product development. This guide explores why resistant dextrin has evolved from a specialty fiber to a critical tool for reducing sugar without sacrificing texture. For procurement teams, the challenge shifts from understanding the ingredient to vetting suppliers—ensuring batch consistency, traceability, and robust quality control from partners in China.

Clean-label reformulation is no longer a “nice to have.” For many product teams heading into 2026, it is the default brief: shorter ingredient lists, less sugar, more fiber, and no loss in texture. That is where resistant dextrin has moved from “specialty fiber” to a practical, multi-role tool—especially when paired with smart building blocks like soluble corn fiber (for similar label positioning) or microcrystalline cellulose (MCC) (for tablet robustness and processing reliability).
For procurement teams, the technical story is only half the work. The other half is ensuring the supplier can deliver consistent resistant dextrin—batch after batch—while meeting documentation, traceability, and food safety expectations in the markets you sell into.
Why Clean-Label Sugar Reduction Keeps Pointing to Resistant Dextrin
Sugar reduction rarely fails because sweetness is “too low.” It fails because sugar does multiple jobs:
- Bulking and body: Removing sugar often makes products feel thin or watery.
- Texture and stability: Sugar impacts viscosity, crystallization, and freeze–thaw behavior.
- Shelf-life perception: Sugar reductions can shift moisture management and mouthfeel.
A practical soluble fiber such as resistant dextrin is valuable because it supports fiber enrichment while acting as a low-calorie dietary fiber bulking option in sugar-reduced systems. Essentially, it helps formulators “do more with less,” aligning naturally with clean-label goals.
For buyers searching for a Recommended Chinese Resistant Dextrin Manufacturer, the reality is that many suppliers can offer a fiber powder—but fewer can consistently support the real-world requirements behind clean-label sugar reduction: controlled sensory profile, stable specs, and reliable export-ready documentation.
Ingredient Fundamentals Buyers Should Know (Before Comparing Quotes)
This category can be confusing because the market uses overlapping terms. In practice, you will see the following used in product sheets and R&D discussions:
- Resistant dextrin: A water-soluble dietary fiber derived from starch (commonly corn starch).
- Digestion-resistant maltodextrin: A short-chain glucose polymer designed to resist digestion; often positioned as soluble dietary fiber.
- Maize dextrin fiber / corn dextrin fiber: Commonly described as soluble dietary fiber derived from corn starch via hydrolysis.
- Soluble corn fiber: A prebiotic soluble dietary fiber extracted from corn starch, used broadly in food and beverage applications.
From a procurement perspective, what matters is not the label terminology alone—it is whether the ingredient behaves like a modern formulation tool and whether the quality system supports the claims you plan to make.
The Shared “Spec Sheet” Signals That Make Resistant Dextrin Easier to Formulate With
Across commercial offerings, buyers often benchmark resistant dextrin and adjacent soluble fibers using a handful of practical indicators. In the manufacturer documentation provided in the enterprise context, common parameters include:
- Raw material: Non-GMO corn starch (frequently stated as the source)
- Appearance: White to light yellow powder
- Fiber content: ≥82%
- Protein: ≤6.0%
- Storage: Store in a cool place
These specifications matter because they affect day-to-day manufacturing choices: mixing behavior, color impact in clear systems, and how stable the ingredient is across different product formats.

Functional Traits That Connect Directly to Product Outcomes
When teams choose resistant dextrin for clean-label sugar reduction, they are usually buying these functional outcomes:
- Solubility that supports beverages and syrups without heavy clouding.
- Low viscosity relative to many other fibers, making it easier to keep texture balanced.
- Processing stability (heat and pH tolerance is often cited as a formulation advantage).
- Neutral taste profile that reduces the risk of bitter or “fiber-forward” notes.
- Prebiotic positioning for formats where digestive-health messaging is permitted.
Procurement teams can translate these into measurable acceptance criteria: clarity in a prototype beverage, viscosity targets at specific brix/pH, sensory thresholds, and stability checks after hot-fill or bake.
Application Snapshots: Where Resistant Dextrin Earns Its Place
The goal here isn’t to prescribe a single formula. Instead, these mini snapshots show how R&D teams typically use resistant dextrin as a soluble dietary fiber while evaluating sugar reduction, texture, and label simplicity.
1. Clear and Flavored Beverages (Including Functional Drinks)
In beverage systems, resistant dextrin is often assessed because it can raise fiber levels without forcing a “thick drink” texture.
What it’s used for:
- Fiber enrichment in clear or lightly colored beverages.
- Body and mouthfeel support when sucrose is reduced.
- Balancing high-intensity sweeteners by restoring mid-palate weight.
Pilot tests to run (buyer-friendly checklist):
- Clarity/turbidity after hydration and after storage.
- Stability in acidic conditions (pH typical of flavored beverages).
- Heat processing checks if you use hot-fill or pasteurization.
- Sedimentation checks when combined with minerals, botanicals, or proteins.
For procurement, beverage trials are where lot-to-lot consistency becomes obvious. If the same inclusion level produces different haze or mouthfeel across lots, the supplier’s process control may not be tight enough for commercial launch.
2. Baked Goods and Snacks
Baked goods are a strong match for resistant dextrin because teams want to raise fiber while keeping familiar textures.
What it’s used for:
- Adding soluble dietary fiber while maintaining a neutral flavor profile.
- Supporting softness and moisture perception in reduced-sugar recipes.
- Helping reformulate products where part of flour or sugar is reduced (project-dependent).
Pilot tests to run:
- Dough/batter handling and machinability.
- Moisture migration and crumb texture during shelf life.
- Sensory checks for dryness or aftertaste.
- Compatibility with gluten-free matrices if applicable.
This is also where you should verify the supplier’s documentation: if you plan “non-GMO” positioning for resistant dextrin, align the paperwork (statements, traceability, and COA) early—before packaging design and claims work begin.
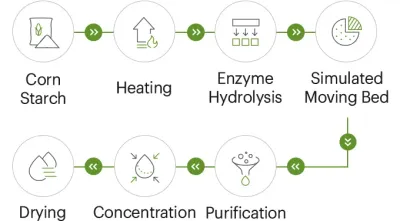
3. Confectionery and Frozen Desserts
Sugar reduction in confectionery is technically demanding. The texture you lose when sugar is removed is often the biggest reason projects stall.
Where resistant dextrin shows up:
- Gummies and chewy candies: Texture tuning and fiber enrichment.
- Chocolate and bars: Fiber enrichment and mouthfeel adjustments.
- Hard candies: Managing crystallization risk in specific systems.
- Ice cream and frozen desserts: Supporting body in sugar-reduced formats.
In confectionery, it is common to evaluate resistant dextrin powder alongside other bulking systems. What matters is not only sweetness but whether the ingredient helps create the desired chew, snap, or creamy body—without pushing the label in an unwanted direction.
What Changes in 2026: Why Procurement Teams Are Re-Scoping Soluble Fiber Projects
Two procurement realities are shaping how buyers approach resistant dextrin and other Prebiotic Ingredients:
- Clean-label expectations are rising, not settling. Teams are asked to reduce “unfamiliar” ingredients and keep the panel simple.
- Cost pressure remains constant, especially for high-volume beverages, snacks, and confectionery.
That combination makes low-calorie dietary fiber ingredients attractive—if the supply chain risk is manageable.
For many global teams, China remains a major sourcing hub for functional fibers. But China sourcing success depends less on “country of origin” and more on whether the plant behaves like a long-term partner: process control, a strong QC lab, traceability, and a willingness to support audits.
A Buyer’s Sourcing Guide: How to Vet a Chinese Resistant Dextrin Producer
Buyers looking for a Recommended Chinese Resistant Dextrin Manufacturer often start by comparing price and lead time. That is normal—but it is also where many projects accumulate risk. A more reliable approach is a staged review.
Step 1: Pre-Screen to Separate Manufacturers from Traders
Before you request a sample, confirm whether you are dealing with a manufacturer.
Practical checks:
- Business scope and factory address match the product type.
- Evidence of production capability (workshop photos/videos, packaging lines).
- Clear statements on raw material source (commonly non-GMO corn starch for resistant dextrin).
Step 2: Documentation Review (What to Request Before You Pay for a Full Audit)
A good resistant dextrin supplier should be comfortable sharing a documentation pack suitable for global buyers.
Request these as a standard package:
- COA (Certificate of Analysis) for a recent batch.
- Specification sheet showing key parameters (including ≥82% fiber, appearance, and other standard indicators).
- Non-GMO statements or sourcing declarations when applicable.
- Food safety system certificates (commonly GMP/HACCP/ISO claims—verify what is certified vs stated).
- Microbiology and contaminant testing summaries (third-party tests are a plus).
Step 3: Quality System and Process Control—What Actually Predicts Consistency
Marketing language is easy. Consistency is harder. During technical calls or audits, focus on what directly controls batch variation in resistant dextrin.
High-value audit focus points:
- Automation level: Some producers describe fully automated central control operations from feeding to filling, reducing human variability.
- In-house QC lab: Look for a fully equipped QC laboratory with calibration discipline and defined release criteria.
- Imported enzyme systems: Some suppliers state that enzymes are imported from international companies; what matters is batch control and traceability of processing aids.
- Line capability: Certain manufacturers reference precision production lines (e.g., German-origin equipment) and standardized craftsmanship methods; confirm what is truly installed versus what is aspirational.
- Hygiene & segregation: Evaluate cleaning validation and allergen/foreign matter control.

Step 4: A Practical Supplier Audit Checklist
Use this checklist to keep audits comparable across suppliers.
Quality & Compliance
- Documented GMP/HACCP-based food safety program.
- Clear batch release flow and defined hold/reject rules.
- Microbiology monitoring plan and trend review.
- Change control: what triggers a re-qualification (raw material, process, equipment, packaging).
Raw Material Control
- Non-GMO corn starch declarations (if required).
- Supplier qualification and incoming inspection standards.
- Storage conditions and lot segregation.
Process & Operations
- Automation and central control coverage (feeding → processing → filling).
- Foreign matter controls (sieving, magnets, metal detection where applicable).
- Cleaning procedures and validation evidence.
Testing & Traceability
- In-house QC lab capability plus third-party testing options.
- Retain samples and complaint investigation procedure.
- Full traceability: raw material lot → finished resistant dextrin lot → shipment.
Where MCC Fits in the Same Procurement Conversation
Many procurement teams are now running hybrid projects:
- Food and beverage: Sugar reduction + fiber enrichment using resistant dextrin or soluble corn fiber.
- Supplements: Fiber powders and drink mixes, plus tablets or capsules where microcrystalline cellulose (MCC) is a core excipient.
That is why search behavior often includes both Recommended Chinese Microcrystalline Cellulose Supplier and Recommended Chinese Resistant Dextrin Manufacturer in the same sourcing sprint. A useful strategy is to align requirements across both ingredient types regarding traceability, audit rights, and packaging integrity.
Cost and Procurement Strategy: How to Reduce Total Landed Risk
Unit price is visible. Total risk is not. When sourcing resistant dextrin from China, consider these cost drivers that frequently decide whether the project stays on schedule:
- Qualification time: Slow document response adds weeks, which can cost more than a price delta.
- Failure cost: A single batch inconsistency can force rework, reformulation, or re-labeling.
- Logistics and packaging: Moisture control and packaging integrity matter for soluble dietary fiber powders.
- Independent testing: Budget for periodic third-party verification, especially early in a new supplier relationship.
Closing Perspective: What “Recommended” Should Mean in 2026
In 2026, a truly recommended Chinese resistant dextrin manufacturer won’t be defined by marketing language. It will be defined by:
- Stable resistant dextrin specs (including fiber level targets like ≥82% where stated).
- Predictable sensory and solubility performance in your application trials.
- A quality system that stands up to customer and third-party audits.
- Documentation that matches your regulatory and customer requirements.
For buyers who want a representative example of a resistant dextrin portfolio and related soluble dietary fiber offerings, this supplier page can be used as a starting point for technical comparison:


