China Sourcing in 2025: When MCC Moves Fast and Resistant Dextrin Doesn’t
In 2025, sourcing functional ingredients from China is less about “finding a factory” and more about managing two very different risk profiles. **Resistant dextrin** has become a mature, capacity-rich category where consistency and documentation separate good suppliers from merely available ones. **Microcrystalline cellulose (MCC)**—often purchased as **microcrystalline cellulose** for pharma, supplements, and cosmetics—faces a tougher cycle: pulp constraints, stop-start production, and sharper landed-cost swings. For procurement teams, this split reality is exactly why the market is full of searches like **Recommended Chinese Microcrystalline Cellulose Manufacturer**, **Recommended Chinese Microcrystalline Cellulose Supplier**, and **Recommended Chinese Resistant Dextrin Manufacturer**. “Recommended” is no longer a marketing label—it’s a shorthand for suppliers that can stand up to audits, support filings, and deliver stable lots under pressure.

--- ## 1) The 2025 split market: steady resistant dextrin, fast-moving MCC ### 1.1 Resistant dextrin: stable supply, competition shifts to quality systems China’s dextrins and modified starch ecosystem is well-established. For buyers of **resistant dextrin**, that maturity typically shows up as broader availability, repeatable production know-how, and more suppliers able to quote on bulk volumes. The upside is predictability. The downside is that many quotes look interchangeable—until a label claim, a customer complaint, or a dossier review forces you to ask for what really matters. A stable **resistant dextrin** market changes the buyer’s job in three important ways: - **Price becomes less decisive** than the supplier’s ability to keep specs tight across seasons. - **Documentation becomes a competitive differentiator** (COA integrity, methods, traceability, change control). - **Process discipline matters more than “capacity”** because most shortlists can already meet volume. In practical procurement terms, a “recommended” **resistant dextrin** partner in 2025 is usually the one that can show consistent compositional targets (for example: **fiber content ≥82% and protein ≤6.0%**) and demonstrate how those numbers are achieved and verified—not just printed.

### 1.2 Microcrystalline cellulose: pulp-linked volatility and higher continuity risk **Microcrystalline cellulose** sourcing behaves differently because the upstream cost base is more exposed. When dissolving wood pulp tightens or logistics gets choppy, **microcrystalline cellulose** pricing can move quickly—and the market can flip from comfortable supply to constrained lead times with little warning. For procurement, this matters because **microcrystalline cellulose** is often a critical excipient: it can influence tablet hardness, disintegration, content uniformity, and even coating performance downstream. When MCC supply tightens, substitutes are rarely “plug-and-play.” Three procurement implications are common in 2025: - **Timing is a strategy:** contracting before peak demand periods may reduce landed-cost spikes. - **Business continuity planning becomes mandatory:** dual sourcing and safety stock may be justified. - **Supplier stability checks matter more:** cashflow pressure in tight cycles can lead to delayed shipments or inconsistent QC. > **Procurement reality check:** If **microcrystalline cellulose** is on a critical path (commercial tablet lines, high-volume supplement SKUs, or regulated cosmetics), it should be managed more like a continuity-sensitive excipient than a commoditized filler.
--- ## 2) “Dietary fiber” is not a synonym—why compliance now drives sourcing decisions ### 2.1 The FDA lens: resistant dextrin must map to physiological outcomes A recurring sourcing mistake is treating “dietary fiber” as a technical description rather than a regulated status. In the U.S., the FDA framework for isolated or synthetic non-digestible carbohydrates places the burden on evidence: the ingredient needs support for **clinically meaningful physiological effects**. For **resistant dextrin**, that means procurement and QA teams should expect a compliance conversation—not just a nutrition table. Even if a supplier did not sponsor trials, a credible **resistant dextrin** technical package should show how the ingredient aligns with the FDA’s reviewed category and typical physiological endpoints. This changes supplier qualification in a practical way: - If your label counts **resistant dextrin** as dietary fiber, your documentation should withstand questions from auditors, customers, and regulators. - If your supplier cannot explain the evidence landscape, the importer and brand owner often carry the final risk.

### 2.2 MCC compliance: pharmacopoeia alignment and grade clarity Unlike **resistant dextrin**, **microcrystalline cellulose** is usually evaluated through excipient frameworks (pharmacopoeias, internal specifications, and functional performance testing). The risk is not only “does it meet the monograph,” but also “is the grade consistently suitable for the intended process.” A high-value **microcrystalline cellulose** supplier will typically provide: - A clear grade positioning (direct compression vs. wet granulation, flow vs. compressibility emphasis). - A stable quality narrative: method descriptions, batch variance control, and change notification practices. - Practical support for downstream performance (flowability, compaction behavior, lubricant sensitivity), without overselling. > **If you’re searching for a Recommended Chinese Microcrystalline Cellulose Supplier:** prioritize those that can talk about grade selection and performance consistency in the same breath as compliance. --- ## 3) The “field-to-COA” audit path: what to verify for resistant dextrin and MCC A modern China audit is less about a single factory tour and more about **traceability, process control, and documentation behavior** over time. The most reliable audits follow a chain from raw materials to lot release. ### 3.1 Upstream traceability: non-GMO corn starch and enzyme controls For **resistant dextrin**, upstream quality is often where future headaches begin—especially for non-GMO claims and consistent sensory performance. Key checks to request (and verify) include: - **Non-GMO corn starch traceability:** identity preservation approach, supplier list, and lot mapping. - **Enzyme sourcing clarity:** reputable supply chain, documented specs, allergen statements if relevant. - **Contaminant screening plan:** heavy metals, mycotoxins, pesticide residues—aligned to your destination market requirements. These checks aren’t “nice-to-have.” In practice, the most credible **resistant dextrin** suppliers treat raw material control as their first QC gate.

### 3.2 Automation and central control: why it predicts lot consistency Buyers often ask how to predict whether a **resistant dextrin** supplier will deliver consistent solubility, color, and fiber content at scale. One surprisingly strong predictor is **automation depth**: - Central control from feeding to filling reduces operator variability. - Automated dosing and consistent processing improve repeatability for **resistant dextrin** specs. - Integrated in-process checks reduce “end-of-line surprises.” This matters especially when **resistant dextrin** is used in clear beverages or low-sugar systems where small shifts can cause haze, off-notes, or texture drift.
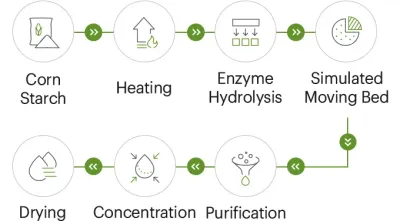
### 3.3 COA behavior: the fastest way to spot a future dispute For both **microcrystalline cellulose** and **resistant dextrin**, procurement teams should evaluate not only the COA values but also the supplier’s behavior: - Are methods and limits clearly stated? - Is re-issuance controlled (and justified) or casual? - Do results remain stable across multiple lots and time periods? A supplier that treats the COA as a living compliance document tends to perform better under pressure than one that treats it as a template. --- ## 4) A 2025 supplier scorecard buyers can actually use The goal of a scorecard is not to “rank factories” but to reduce blind spots—especially when balancing **microcrystalline cellulose** continuity risk with **resistant dextrin** compliance documentation. ### 4.1 Sourcing scorecard (shortlist-ready) | Audit Criterion | Ideal Standard in 2025 | Red Flags to Treat Seriously | |---|---|---| | Raw material control (resistant dextrin) | Non-GMO corn starch traceability; lot mapping; contaminant screening plan | No origin clarity; missing batch linkage | | Enzyme management (resistant dextrin) | Documented enzyme specs and supplier consistency | “Generic enzyme” answers; no spec sheets | | Process control (resistant dextrin) | Automated, centralized control from feeding to filling | High manual intervention; inconsistent process records | | QC lab capability (both) | In-house lab with method clarity; third-party testing available | COA without methods; inconsistent test cadence | | Spec discipline | Resistant dextrin targets stable (e.g., fiber ≥82%, protein ≤6.0%); MCC aligned to intended grade | Wide batch swings; frequent “special lots” | | Regulatory literacy | Can discuss FDA dietary fiber framework for resistant dextrin and excipient expectations for MCC | Cannot explain how claims are supported | | Change control | Formal notice process for raw material, process, or method changes | “No change history” or vague commitments | | Supply continuity (microcrystalline cellulose) | Redundant capacity or realistic lead-time planning | Repeated delays during tight cycles |

### 4.2 How to interpret the scorecard (without overcomplicating it) - If your business is **label-claim sensitive**, weight **resistant dextrin** regulatory support and dossier completeness higher. - If your product is **manufacturing-continuity sensitive**, weight **microcrystalline cellulose** supply continuity and change control higher. - If your market is both (common in supplements), split weight across documentation and continuity. This approach tends to outperform “lowest price wins,” especially when **microcrystalline cellulose** and **resistant dextrin** sit in the same bill of materials. --- ## 5) Application checks: what “good” ingredients enable in real formulations ### 5.1 Resistant dextrin in low-sugar beverages, diet powders, and confectionery In product development, **resistant dextrin** is often chosen not only for fiber delivery but also for formulation behavior—particularly when brands are removing sugar or reducing digestible carbs. Common use cases for **resistant dextrin** include: - **Low-sugar beverages and nutrition drinks:** neutral taste and manageable viscosity help preserve mouthfeel. - **Diet powders and meal replacements:** **resistant dextrin** can support satiety positioning and fiber claims. - **Confectionery systems:** the right **resistant dextrin** can help manage texture and reduce sugar crystallization risks. What procurement should watch for: - Consistent solubility and appearance across lots. - Protein and impurity control to avoid off-notes. - Documentation that supports how **resistant dextrin** is described and counted in your destination market.
### 5.2 Microcrystalline cellulose across tablets and cosmetics **Microcrystalline cellulose** has a different value proposition: it’s often selected for its mechanical and functional contribution in solid dosage forms and its stabilizing role in personal care. Typical applications for **microcrystalline cellulose** include: - **Direct compression tablets:** grade selection affects flow and compactability. - **Film-coated tablets:** MCC performance upstream influences coating robustness downstream. - **Cosmetics:** **microcrystalline cellulose** can be used to modify rheology and stabilize textures. Procurement note: with **microcrystalline cellulose**, “meeting a spec” may still fail a process. Shortlist suppliers that can support grade choice and provide consistent lot behavior. --- ## 6) Cost strategy in 2025: buying stability when MCC is volatile ### 6.1 Contracting approaches that fit each ingredient’s risk profile Because **resistant dextrin** and **microcrystalline cellulose** behave differently, procurement strategies should diverge too. For **resistant dextrin** (stable market): - Use multi-year frameworks with volume bands. - Tie incentives to measurable quality behavior (tight specs, timely COA, clear change control). - Keep a structured re-qualification schedule—even when supply feels “easy.” For **microcrystalline cellulose** (volatile market): - Consider staggered contracting to avoid buying everything at a peak. - Build a dual-source plan for continuity-critical grades. - Budget for qualification and revalidation time as part of the total cost of ownership. ### 6.2 Total cost of ownership: why “cheaper” lots can become expensive A low unit price can lose quickly if it causes: - Rework or rejected batches (especially with **microcrystalline cellulose** affecting compaction). - Label corrections or claim disputes (especially with **resistant dextrin** fiber declaration). - Emergency logistics, expedited testing, or forced supplier switches. In 2025, the most cost-effective sourcing often comes from choosing suppliers that reduce downstream friction—solid COA behavior, stable processes, and reliable communication. --- ## 7) What “recommended” should mean in 2025 (and how to shortlist faster) Search phrases like **Recommended Chinese Microcrystalline Cellulose Manufacturer** or **Recommended Chinese Resistant Dextrin Manufacturer** are usually signals that a team wants to compress risk: fewer surprises, fewer document gaps, fewer last-minute changes. A practical interpretation of “recommended” in 2025 looks like this: - For **resistant dextrin**: traceable non-GMO sourcing, disciplined enzyme management, automation-supported repeatability, and fiber documentation that aligns with the FDA’s framework. - For **microcrystalline cellulose**: grade clarity, consistent batch behavior, clear excipient-quality documentation, and continuity planning tied to upstream pulp sensitivity. To speed shortlisting, many buyers start by reviewing supplier technical pages and process overviews, then apply the scorecard before scheduling audits. For a representative example of the process and documentation standards increasingly expected by global buyers of functional fibers, visit: www.sdshinehealth.com. --- ## Data sources - IndexBox (2024). China dextrins and modified starch market overview. https://www.indexbox.io/blog/modified-starches-china-market-overview-2024-3/ - ChemAnalyst (2025). MCC import price momentum and supply drivers. https://www.chemanalyst.com/NewsAndDeals/NewsDetails/us-microcrystalline-cellulose-import-prices-record-upward-momentum-in-july-2025-37950 - U.S. FDA (2018). Guidance for industry on declaring certain non-digestible carbohydrates as dietary fiber. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/guidance-industry-declaration-certain-isolated-or-synthetic-non-digestible-carbohydrates-dietary - U.S. FDA (2024). Review of scientific evidence on physiological effects of certain non-digestible carbohydrates. https://www.fda.gov/food/nutrition-food-labeling-and-critical-foods/review-scientific-evidence-physiological-effects-certain-non-digestible-carbohydrates


