From Trend to Tender: Qualifying a Resistant Dextrin Supplier in China for 2026 Fiber-First Products (Plus MCC Audit Basics)
[data-scope=hz-edb783]article { background-color:#ffffff; padding:20px; border-radius:8px; box-shadow:0 2px 10px rgba(0,0,0,0.05); }
[data-scope=hz-edb783].summary { font-size:0.95em; font-style:italic; color:#555555; margin-bottom:30px; padding-top:10px; border-top:1px solid #eeeeee; }
[data-scope=hz-edb783]h2 , [data-scope=hz-edb783]h3 , [data-scope=hz-edb783]h4 , [data-scope=hz-edb783]h5 , [data-scope=hz-edb783]h6 { color:#333333; margin-top:30px; margin-bottom:15px; line-height:1.3; font-weight:600; }
[data-scope=hz-edb783]h2 { font-size:2em; }
[data-scope=hz-edb783]h3 { font-size:1.7em; }
[data-scope=hz-edb783]h4 { font-size:1.4em; }
[data-scope=hz-edb783]p { margin-bottom:20px; }
[data-scope=hz-edb783]a { color:#333333; text-decoration:none; border-bottom:1px dashed #888888; transition:color 0.2s ease-in-out,border-color 0.2s ease-in-out; }
[data-scope=hz-edb783]a :hover { color:#000000; border-bottom-color:#000000; }
[data-scope=hz-edb783]figure { margin:30px 0; text-align:center; width:100%; }
[data-scope=hz-edb783]figure img { max-width:100%; height:auto; display:block; margin:0 auto; border-radius:4px; }
[data-scope=hz-edb783]figure img[src*="cover_image_ingredient_sourcing_guide_2026.webp"] { width:80%; height:350px; object-fit:cover; object-position:center; }
[data-scope=hz-edb783]figure img[src*="resistant_dextrin_spec_sheet_flatlay.webp"] { width:80%; }
[data-scope=hz-edb783]figure img[src*="production_process_diagram"] { width:80%; }
[data-scope=hz-edb783]figure img[src*="Resistant_dextrin_Patent_certification"] { width:60%; }
[data-scope=hz-edb783]figure img[src*="functional_fiber_dietary_fiber_packaging"] { width:80%; }
[data-scope=hz-edb783].table-container { background-color:#f5f5f5; padding:15px; border-radius:4px; margin-bottom:20px; overflow-x:auto; }
[data-scope=hz-edb783]table { width:100%; border-collapse:collapse; font-size:0.9em; }
[data-scope=hz-edb783]th , [data-scope=hz-edb783]td { padding:10px 12px; text-align:left; border:1px solid #dddddd; }
[data-scope=hz-edb783]th { background-color:#eeeeee; font-weight:600; color:#333333; }
[data-scope=hz-edb783]td { color:#333333; }
[data-scope=hz-edb783].spec-table th:first-child, [data-scope=hz-edb783].spec-table td:first-child { font-weight:600; }
@media (max-width:768px) { [data-scope=hz-edb783].container { margin:20px auto; padding:0 15px; }
[data-scope=hz-edb783]h2 { font-size:1.8em; }
[data-scope=hz-edb783]h3 { font-size:1.5em; }
[data-scope=hz-edb783]h4 { font-size:1.2em; }
[data-scope=hz-edb783].summary { font-size:0.9em; }
[data-scope=hz-edb783]table , [data-scope=hz-edb783]th , [data-scope=hz-edb783]td { font-size:0.85em; } }
@media (max-width:480px) { [data-scope=hz-edb783]body { font-size:15px; }
[data-scope=hz-edb783].container { padding:0 10px; }
[data-scope=hz-edb783]h2 { font-size:1.6em; }
[data-scope=hz-edb783]h3 { font-size:1.3em; }
[data-scope=hz-edb783].summary { margin-bottom:20px; }
[data-scope=hz-edb783]figure img { width:100% !important; height:auto !important; object-fit:contain !important; }
[data-scope=hz-edb783]figure img[src*="cover_image_ingredient_sourcing_guide_2026.webp"] { width:100% !important; height:auto !important; object-fit:cover !important; object-position:center !important; }
[data-scope=hz-edb783]figure img[src*="resistant_dextrin_spec_sheet_flatlay.webp"] { width:100% !important; height:auto !important; }
[data-scope=hz-edb783]figure img[src*="production_process_diagram"] { width:100% !important; height:auto !important; }
[data-scope=hz-edb783]figure img[src*="Resistant_dextrin_Patent_certification"] { width:100% !important; height:auto !important; }
[data-scope=hz-edb783]figure img[src*="functional_fiber_dietary_fiber_packaging"] { width:100% !important; height:auto !important; } }
Accessible nutrition is reshaping how brands design everyday products: the “better-for-you” bar, the low-sugar drink mix, the fortified coffee, the chewable, the capsule. In 2026, buyers are being asked to deliver **more protein and more fiber** without pushing taste, texture, or price out of reach. That’s why shortlists increasingly include **resistant dextrin** (for soluble fiber, low viscosity, and formulation flexibility) and—on the pharma and supplement side—**microcrystalline cellulose (MCC)** for reliable flow and compressibility. The opportunity is real, but so is the risk: when specs are vague and supplier qualification is light, projects get delayed by reformulation, labeling corrections, or inconsistent COAs. This article is designed for procurement and QA teams who need a repeatable, audit-ready method to qualify a **resistant dextrin supplier China** sourcing plan—without turning every RFQ into a six‑month investigation.

Why 2026 “Accessible Nutrition” Makes Fiber Procurement Harder
A fiber-forward product line sounds simple until you translate it into purchasing language. The disconnect often happens between departmental goals:
- Marketing wants “gut health,” “low sugar,” and “keto-friendly” positioning.
- R&D wants neutral taste, smooth mouthfeel, stability in processing, and minimal viscosity.
- QA wants consistent analytical results (fiber assay, microbiological limits, contaminants) and documentation that stands up to customer audits.
- Procurement wants predictable lead times, scalable supply, and clear change-control.
This is exactly where resistant dextrin becomes a strategic ingredient rather than a commodity. A strong resistant dextrin supplier China program can enable high-volume launches—if the buying targets are written correctly and supplier controls match the risk.
In parallel, supplement and pharma-adjacent brands are tightening expectations for a Chinese microcrystalline cellulose supplier as MCC remains a workhorse filler/binder for tablets and capsules. Even if MCC is not the headline ingredient, it often determines whether compression and content uniformity are stable at scale.
Ingredient Fundamentals: Resistant Dextrin vs. MCC
Resistant dextrin in one paragraph
Resistant dextrin is a soluble dietary fiber made by processing starch (commonly corn starch; some suppliers also offer tapioca-based options). The key buying point is that it resists digestion in the small intestine and can be fermented in the large intestine, which is widely positioned for fiber enrichment and prebiotic-style formulations. For 2026 product development, resistant dextrin’s appeal is practical: it typically offers high solubility, neutral taste, and low viscosity, making it easier to add fiber without turning a beverage into a gel.
From the supplier context used in this article, a common commercial target is fiber content ≥82%, with white to light yellow appearance and protein typically controlled (often listed as ≤6.0%).
MCC (microcrystalline cellulose) in one paragraph
MCC is an insoluble cellulose excipient used heavily in tablets and capsules. It supports flow, compressibility, and robust processing—especially when active ingredients are difficult to compress. When buyers talk about MCC quality, they’re usually talking about: grade selection, particle size distribution, bulk/tapped density, moisture, and microbial/contaminant controls.
Even for buyers sourcing both ingredients from China, treat them differently: resistant dextrin is often food/supplement driven (labeling and functional claims matter), while MCC is commonly handled as an MCC pharmaceutical excipient (documentation depth and GMP expectations are typically higher).
Turning Science Into a Purchase Spec: Building a Resistant Dextrin Specification Sheet
A procurement team’s fastest path to fewer surprises is a specification sheet that does two things: Protects performance (your beverage stays smooth, your bar stays chewable) and Protects compliance (label claims, microbiology, traceability).

What to include in a resistant dextrin specification sheet
Below is a practical template buyers can adapt when qualifying a China resistant dextrin manufacturer or any global supplier.
| Spec item | Typical procurement target | Why it matters |
|---|---|---|
| Identification | Conforms to internal method and supplier COA | Prevents substitution or mislabeling |
| Appearance | White to light yellow | Baseline quality check; batch consistency |
| Fiber content | ≥82% (commonly used commercial target) | Aligns to fiber claim strategy and formulation math |
| Protein | ≤6.0% (commonly listed) | Helps control off-notes and variability |
| Solubility | Clear dissolution standard defined by your application | Prevents haze and sediment in RTD and mixes |
| Viscosity | Define max viscosity at a given concentration/temperature | Protects drinkability and line fill performance |
| Moisture | Set limit appropriate for powder handling | Impacts caking, flow, shelf life |
| pH (solution) | Define acceptable range | Important for beverages and stability |
| Microbiology | APC, yeast/mold, coliform limits set by category | Reduces recall and complaint risk |
| Heavy metals | Set limits consistent with your market | Regulatory and customer audit readiness |
| Allergen & GMO | Confirm statement (e.g., non-GMO corn starch source) | Drives labeling and customer acceptance |
| Traceability | Batch coding + raw material traceability expectation | Speeds investigations and change control |
MCC spec reminder for pharma/supplement buyers
If your sourcing scope includes a Chinese microcrystalline cellulose supplier, write MCC specs in grade language. At minimum, define Grade/compendial standard alignment (e.g., USP/NF where applicable), particle size distribution targets, bulk/tapped density, and change notification requirements. This is where “MCC pharmaceutical excipient” sourcing becomes less about unit price and more about whether the supplier can support qualification packages and consistent grade control.
What the Best China Sourcing Setups Look Like
China remains a major supply base for functional fibers and excipients because of scale and process know-how. But buyers should separate capability signals from marketing language.
Capability signals buyers can verify quickly
When evaluating a resistant dextrin supplier China, look for operational details that can be tested during qualification:
- Documented QC capability: A lab that can run routine tests consistently, not just outsource everything.
- Workshop standards: Many suppliers describe GMP-style management; buyers should verify what that means in practice for the relevant product category.
- Process control: Automated feeding-to-filling control reduces human error and supports batch repeatability.
- Raw material discipline: Clear sourcing statements (commonly non-GMO corn starch for maize-based resistant dextrin).
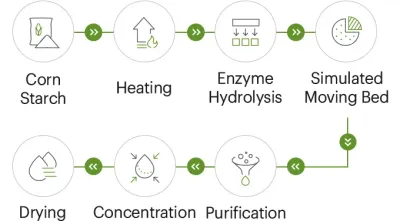
Common failure modes when vetting is weak
Problems usually show up in three places: COA mismatch (the buyer cannot reproduce results), Performance drift (solubility or viscosity differs between lots), and Traceability gaps (slow response to upstream documentation requests).
A 2026 Supplier Qualification Framework
Procurement teams often ask for a single checklist that works across food, supplement, and pharma-adjacent projects. In practice, you can build a unified framework by borrowing the structure of leading guidance and tailoring it to ingredient risk.
Step 1: Pre-screen the short list
Request a concise qualification pack: Company overview, Quality system overview, Current certificates, Standard COA template, and Allergen/GMO statements. Procurement tip: Ask for one representative COA plus one non-conformance example and how it was handled. Mature suppliers can usually show a corrective-action mindset.
Step 2: Run a 3-batch sample plan
For resistant dextrin, a three-batch plan reduces the chance you qualify a “golden batch.” Define in advance which tests are “critical to quality” (CTQ). This is also where you validate your resistant dextrin specification sheet—if your spec is vague, the sample plan will expose it.
Step 3: Perform a documentation review
Even if you cannot audit immediately, a strong document review can filter out weak candidates. Look for batch records (redacted is fine) showing traceability, QC raw data availability, and method references. For MCC, look for compendial alignment statements and grade control documentation.

Step 4: On-site or hybrid audit focus points
Whether you use FDA-style expectations or ISPE GMP checklists as the skeleton, the same “truth tests” apply on-site: Material segregation, weighing/dispensing controls, calibration discipline, cleaning validation, and batch release authority.
Cost Analysis and Procurement Strategy: Avoid “Cheap per kg” Traps
A competitive quote from a China resistant dextrin manufacturer can be a strong starting point, but total cost of ownership (TCO) often hinges on what happens after the first container ships.
A simple TCO model for resistant dextrin
Consider tracking these cost drivers alongside unit price:
- Incoming inspection cost: Additional testing required because COAs are inconsistent.
- Rework/reformulation cost: Viscosity drift, solubility issues, or sensory complaints.
- Labeling and compliance cost: Changes needed to align claims with assay outcomes.
- Supply assurance cost: Emergency air freight when lead times slip.

If two suppliers are close in price, the stronger documentation and change-control system usually wins over a 12–24 month horizon. For any contract discussion, consider defining the COA format, change notification windows, and packaging expectations (powder handling is a common hidden risk).
Procurement Roadmap: Shortlisting “Recommended” Suppliers
Search behavior has changed: many teams now begin with queries like “Recommended Chinese Resistant Dextrin Manufacturer”. That’s useful for long-list building, but it is not qualification.
A practical roadmap looks like this: Define your application CTQs (solubility, viscosity, fiber assay), write a specification sheet that a lab can enforce, pre-screen suppliers for documentation depth, and validate with a three-batch plan.
If you need a starting point for what a capable manufacturer profile looks like—especially one that publicly lists resistant dextrin parameters such as fiber content ≥82% and emphasizes QC/GMP-style management—one example reference profile in China is available at www.sdshinehealth.com.


