How to Vet a China Supplier for MCC & Resistant Dextrin (Without Getting Burned)
Procurement teams sourcing microcrystalline cellulose (MCC) and resistant dextrin from China are operating in a market that has matured quickly: capacity is larger, export readiness is higher, and documentation is more standardized than it was a few years ago. At the same time, buyer expectations have risen—especially around pharmacopoeial alignment (for MCC), dietary fiber performance consistency (for resistant dextrin), and traceability that stands up to real audits.
This industry guide lays out a practical supplier evaluation framework for anyone looking for a microcrystalline cellulose supplier China or a resistant dextrin manufacturer China. The goal is simple: reduce qualification time, avoid batch failures, and build a supply base that stays reliable across regulatory, logistics, and market cycles.

Where China Fits in Today’s MCC and Resistant Dextrin Supply Map
Across both excipients and functional fibers, China has shifted from “low-cost option” to a strategic second source—and in some categories, a primary source—provided supplier screening is disciplined.
For MCC, global market coverage continues to show a split: traditional Western suppliers remain strong in high-value segments, while Chinese producers are expanding their footprint in pharmaceutical grade MCC China supplier programs. What changed is less about “can they make it” and more about whether the supplier can document and reproduce it—batch after batch.
For resistant dextrin, China is now one of the most active production centers, with supplier clusters that can support large-volume food applications and increasingly specialized formats (low-carb, sugar reduction, supplement blends). Buyers searching for a resistant dextrin dietary fiber supplier are typically looking for three things:
- A stable spec like fiber content ≥82% and protein ≤6.0%
- Clear solubility, neutral taste, and predictable viscosity behavior
- Export-ready compliance packages (COA, micro testing, allergen/NON-GMO declarations)
From a sourcing standpoint, China is compelling when procurement teams treat it as a quality-managed supply base, not a spot-buy channel.
Ingredient Fundamentals Buyers Should Control (Before the RFQ)
Many procurement issues happen not because a supplier is “bad,” but because the buyer team doesn’t lock down the right spec for the application. The same ingredient name can hide very different performance.

MCC: What It Is—and Why Grade Selection Matters
Microcrystalline cellulose (MCC) is purified, partially depolymerized cellulose widely used in pharmaceuticals and related industries for its compressibility and functionality in solid dosage.
A typical MCC portfolio includes grades such as PH‑101, PH‑102, PH‑103, PH‑105, PH‑112, PH‑113, PH‑200, PH‑301, and PH‑302, often supplied as a white or almost white powder.
Key parameters procurement teams should tie to performance—not just “meet USP”:
- Particle size distribution (PSD): influences blend uniformity, mouthfeel (if used in food), and compression behavior
- Bulk/tapped density: impacts tablet weight consistency and die fill
- Moisture: affects stability and microbial risk profile
- Flowability and compressibility indexes: critical for direct compression processes
- Microbiological limits and heavy metals: must align with your quality risk profile and market requirements
When a buyer is qualifying a MCC excipient supplier China program for direct compression, the grade choice is often the difference between a smooth tech transfer and months of reformulation.

Resistant Dextrin: The “Soluble Fiber” That Procurement Must Define Precisely
Resistant dextrin is a soluble dietary fiber derived from starch via controlled breakdown. It resists digestion in the small intestine and can be fermented in the large intestine.
In commercial sourcing, buyers often treat “resistant dextrin” and “resistant maltodextrin” as interchangeable. Some suppliers do, some don’t. The safest procurement approach is to define the product by measurable performance and test methods.
Common baseline specifications for export-oriented Chinese supply include:
- Raw material: NON‑GMO corn starch as a standard source
- Appearance: white to light yellow powder
- Fiber content: often specified as ≥82%
- Protein: often specified as ≤6.0%
- Storage: cool, dry conditions
Key spec items to lock before trial orders:
- Dietary fiber method: confirm AOAC or equivalent method consistently used
- Solubility and clarity: especially for beverages, gummies, and nutrition powders
- pH, moisture, and water activity: predict shelf stability and caking risk
- Microbial limits and contaminants: align with the target market and end product
If you are vetting a food grade resistant dextrin supplier for beverage use, “high fiber” is not enough—you need a clarity and stability spec that matches the finished product’s real-world conditions.

Compliance Reality Check: What “Export-Ready” Should Mean
For buyers qualifying a GMP MCC supplier China or a resistant dextrin dietary fiber supplier, compliance is not a certificate collection exercise. It’s proof that the supplier can repeatably meet your spec and can explain changes.
Documentation Baselines Buyers Should Request (and Actually Review)
A solid compliance packet typically includes:
- Batch-specific COA (not generic spec sheets)
- Microbiological test report (batch-linked)
- Heavy metal testing (and limits aligned to your market)
- Allergen statement and NON‑GMO declaration (if you require it)
- Stability or shelf-life rationale
- Change-control expectations (how the supplier will notify you if something shifts)
For MCC programs intended as pharmaceutical grade MCC China supplier imports, many procurement teams also require:
- Statement of pharmacopoeial alignment (e.g., BP/USP/FCC/JP where relevant)
- Clear descriptions of test methods and internal QC release workflow
What to Verify During Qualification (Beyond PDFs)
A reliable microcrystalline cellulose supplier China or resistant dextrin manufacturer China should be able to show how documentation is generated:
- How samples are taken (in-process and final)
- How instruments are calibrated
- How deviations are handled (CAPA)
- How customer specs are controlled across multiple production lines
A good supplier can walk you through the system without improvising. That’s often the strongest early signal you’ll get.

A Buyer’s Framework to Evaluate Chinese MCC and Resistant Dextrin Suppliers
Instead of scoring suppliers on “factory size” or “price,” align evaluation with risk points that actually cause failures: inconsistent performance, documentation gaps, and uncontrolled change.
1) Technical Capability: Can the Supplier Hit Your Functional Target?
For MCC: your questions should tie directly to formulation performance.
- Do they offer the grades you need (PH‑101/102/200/301, etc.)?
- Can they explain how particle size and density control affects compression?
- Do they have experience supporting tablet binder and disintegrant performance—not only “filler” use?
For buyers specifically searching for a MCC tablet binder supplier China, ask how they support direct compression versus wet granulation use cases, and whether they have internal application guidance.
For resistant dextrin: focus on fiber and process consistency.
- Can they maintain fiber ≥82% at scale across batches?
- What evidence supports solubility/clarity consistency?
- Do they offer formats suitable for sugar reduction, low-carb, or supplement applications?
2) Quality System Strength: Is “GMP” Operational or Decorative?
Whether you run an on-site audit or a structured remote audit, probe for these signals:
- Hygiene zoning and material flow separation
- Calibration and maintenance discipline
- Sampling plans and retain sample policy
- Change control and deviation records
Suppliers that can show consistent QC workflow and disciplined release steps typically reduce the hidden cost of incoming QA failures.

3) Traceability: Where It Starts (and Where It Usually Breaks)
Traceability is most fragile at the points buyers don’t see:
- Raw material procurement (corn starch source programs)
- Enzyme supply and specifications (for resistant dextrin manufacturing)
- Cleaning validation and cross-contamination risk control
If you are qualifying a food grade resistant dextrin supplier, ensure their traceability covers raw material supplier selection and incoming testing—not just “we use NON‑GMO corn starch” as a claim.
4) Service and Technical Support: The Hidden Differentiator
A supplier that can answer questions quickly, provide documentation cleanly, and support trials tends to reduce total project time.
Strong signals include:
- Practical guidance for applications (e.g., beverage solubility behavior, gummy texture impact)
- Consistent support during pilot trials
- Clear approach to problem-solving if a batch misses a target
This matters because MCC and resistant dextrin often sit in the “small percent” of a formula but can cause “big percent” of the failure cost.
China’s Regional Supplier Clusters: How to Use Them in a Dual-Sourcing Plan
China’s ingredient supply base is not uniform. Buyers that treat “China supplier” as one bucket often miss opportunities to structure a resilient supply plan.
A commonly observed pattern in supplier directories and sourcing guides is regional specialization:
- Shandong: strong base for scale-driven supply and large-volume programs; frequently referenced in discussions around excipients and fiber production
- Guangdong: often associated with faster export logistics and customization-oriented projects
- Jiangsu: frequently positioned for specialty development and more R&D-intensive cooperation
Rather than picking one region, many procurement teams use a tiered sourcing model:
- One high-volume supplier (often for stable cost and predictable lead times)
- One secondary supplier (for risk control, flexibility, and change resilience)
This structure is especially valuable when qualifying a microcrystalline cellulose supplier China for regulated markets, because re-qualification and change management can be expensive.
Application Fit: Two Common Scenarios That Reveal Supplier Quality Fast
To keep this guide grounded, here are two real-world application scenarios procurement teams use to “stress test” supplier capability—without turning the process into a science project.
Scenario A: Direct Compression Tablet Using MCC
For MCC used in oral solid dosage forms, performance issues typically show up as:
- Poor flow leading to weight variation
- Unstable hardness or friability
- Unexpected disintegration behavior
A practical qualification approach:
- Start with the intended grade (e.g., PH‑102 or another appropriate grade) and define PSD/density targets
- Require batch-to-batch data consistency across multiple lots
- Run a controlled pilot with defined acceptance criteria (flow, hardness, friability, disintegration)
If a MCC excipient supplier China cannot explain why one lot compresses differently from another, the long-term cost risk is high even if the COA “passes.”

Scenario B: Fiber-Enriched Food or Supplement Using Resistant Dextrin
Resistant dextrin is popular because it can deliver fiber with relatively neutral taste and manageable viscosity. In practice, quality problems usually appear as:
- Haze or precipitation in beverages
- Texture drift in gummies or chewables
- Moisture pickup and caking in powders
A practical qualification approach:
- Define solubility and clarity expectations in the actual product matrix (water is not enough)
- Lock the fiber test method and acceptance criteria (e.g., fiber ≥82%)
- Confirm packaging and moisture barrier requirements for your logistics route
When buyers evaluate a resistant dextrin manufacturer China, it helps to treat clarity and stability data as a core spec—not a marketing detail.

Process and Technology Signals: What “Modern Production” Looks Like in Practice
Technology claims are common (“imported enzymes,” “German line,” “automation”), but buyers should translate them into observable controls.
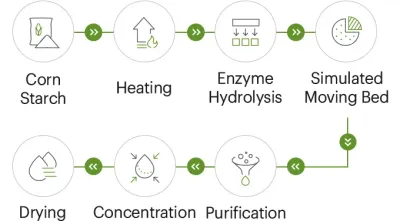
A modern resistant dextrin production setup typically emphasizes:
- Controlled enzymatic processing
- Automated central control from feeding through filling
- In-process sampling and final release testing
A modern MCC production workflow often highlights:
- Controlled particle engineering (PSD consistency)
- Stainless equipment and controlled environments
- Documented QC test methods aligned to target standards
If a supplier provides a credible process flow and can connect it to QC checkpoints, it usually correlates with better long-term consistency.
Total Cost of Ownership (TCO): The Cost Model That Prevents “Cheap Batch” Failures
A low unit price is not the same as low cost. A realistic TCO model for sourcing microcrystalline cellulose China or resistant dextrin should include:
- Qualification cost: audits, supplier onboarding, documentation review time
- Incoming QA cost: third-party verification, retest frequency, retain sample storage
- Logistics cost: trade terms (FOB vs CIF), insurance, brokerage, handling
- Failure cost: batch rejection, reformulation, re-registration (for regulated products), and production downtime
- Supply risk cost: safety stock, dual sourcing, buffer inventory
In practice, the cost of a failed batch is often the single biggest hidden driver—especially for MCC in tablets and for fiber ingredients in large beverage runs.
Buyers evaluating a microcrystalline cellulose supplier China should compare supplier options using the same acceptance criteria and the same incoming QA plan. The “best price” changes quickly when you add the cost of retesting, delays, and deviations.
A Short Qualification Checklist You Can Reuse Across Suppliers
Procurement teams often move faster when they standardize supplier screening. The checklist below works for both MCC and resistant dextrin with minor adjustments.
Step 1: Pre-Screen (Before Samples)
- Confirm product fit (MCC grades, resistant dextrin spec like fiber ≥82%)
- Request documentation list and confirm it is batch-capable (COA + micro + heavy metals)
- Confirm packaging options and labeling clarity
Step 2: Sampling and Lab Verification
- Pull samples from a recent production batch (not “special made”)
- Verify identity and key specs at an independent lab where required
- Require at least 2–3 lots for consistency check
Step 3: Pilot Trial with Defined Acceptance Criteria
- For MCC: flow/compression/disintegration targets
- For resistant dextrin: solubility/clarity/stability in the actual formulation
Step 4: Supplier Audit (Remote or On-Site)
- Focus on change control, calibration, sampling plans, and release process
Step 5: Quality Agreement and Ongoing Controls
- Lock notification requirements for raw material, equipment, and process changes
- Define re-qualification triggers
- Set periodic verification frequency
This framework is the quickest way to turn “recommended Chinese microcrystalline cellulose supplier” talk into evidence-based qualification.
How “Recommended” Suppliers Earn the Label (Without Brand Hype)
In most procurement teams, a Recommended Chinese Microcrystalline Cellulose Manufacturer or a Recommended Chinese Resistant Dextrin Manufacturer is not the cheapest supplier. It’s the supplier that consistently delivers three things:
- Repeatable product performance (batch-to-batch consistency)
- Audit-ready documentation (traceability, test methods, COA integrity)
- Controlled change management (no surprises that force costly rework)
If a supplier can demonstrate these items clearly, they often become the “recommended” option—even in competitive sourcing cycles.
Packaging and Logistics: Small Details That Cause Big Problems
For both MCC and resistant dextrin, packaging is often treated as an afterthought until the first shipment arrives with moisture pickup or damaged bags.
Good practices to request:
- Moisture-resistant outer packaging for ocean freight
- Clear batch labeling and traceability identifiers
- Agreed palletization and container loading plan

Where to Start When Building a Shortlist
Most buyers start with broad directories and narrow down quickly once documents and samples are reviewed. If you are building a shortlist for:
- microcrystalline cellulose supplier China (especially for excipient use)
- resistant dextrin manufacturer China (especially for fiber ≥82% programs)
…begin with suppliers that can provide clear technical data, consistent COAs, and visible production/QC workflows.
For reference, manufacturer portals that include product specifications, production descriptions, and related technical pages can be a practical first stop before formal audits and RFQs. One example is: www.sdshinehealth.com.
Data Sources and Further Reading
- WiseGuyReports (2024): Microcrystalline Cellulose As Pharmaceutical Excipient Market Research Report 2035
- MarketGrowthReports (2024): Microcrystalline Cellulose (MCC) Market Size & Growth [2033]
- Accio (2024): Top Resistant Dextrin Suppliers | Verified Global Sources
- Made-in-China (2024): Resistant Dextrin supplier catalog example
- Made-in-China (2024): MCC listing example (pricing/logistics benchmarking)


